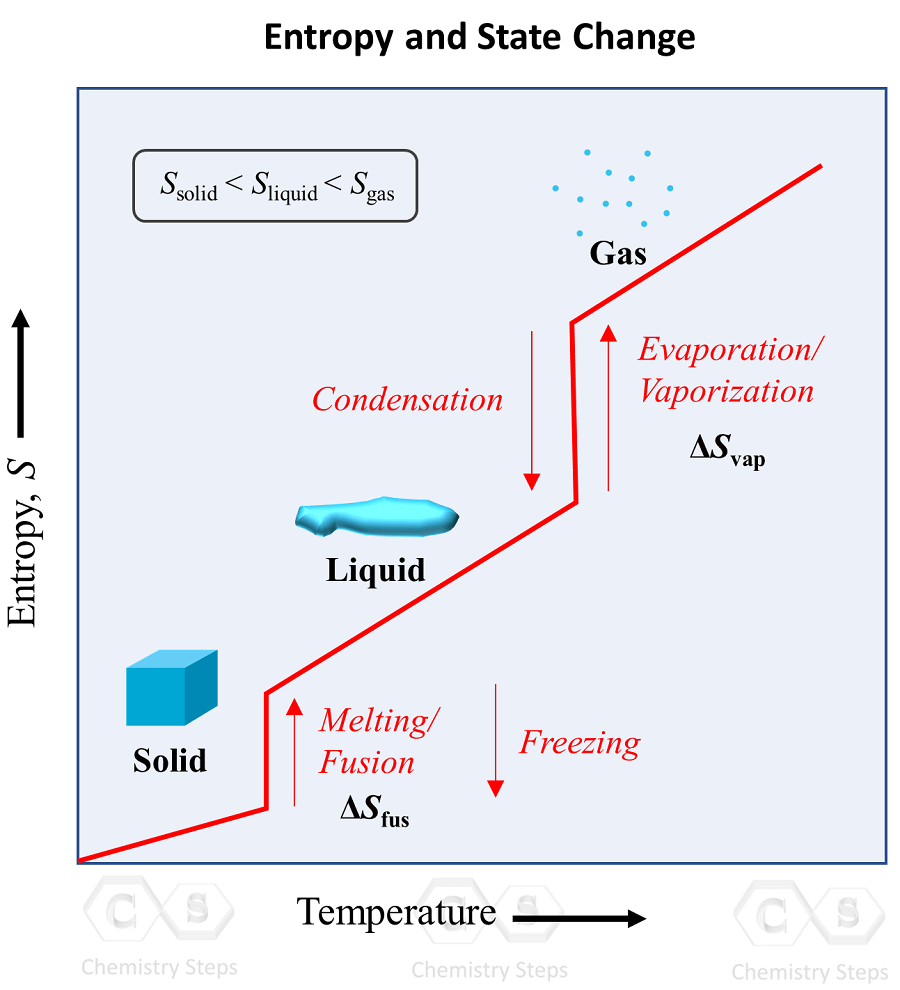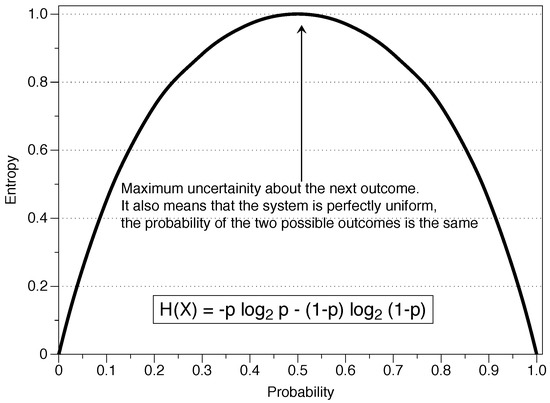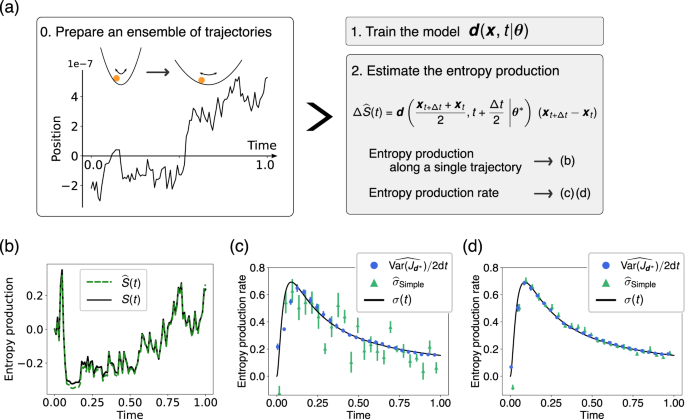
Estimating time-dependent entropy production from non-equilibrium trajectories | Communications Physics

SOLVED:Find the entropy for the following water states and indicate each state on a T-s diagram relative to the two-phase region. a. 250^∘ C, v=0.02 m^3 / kg b. 250^∘ C, 2000

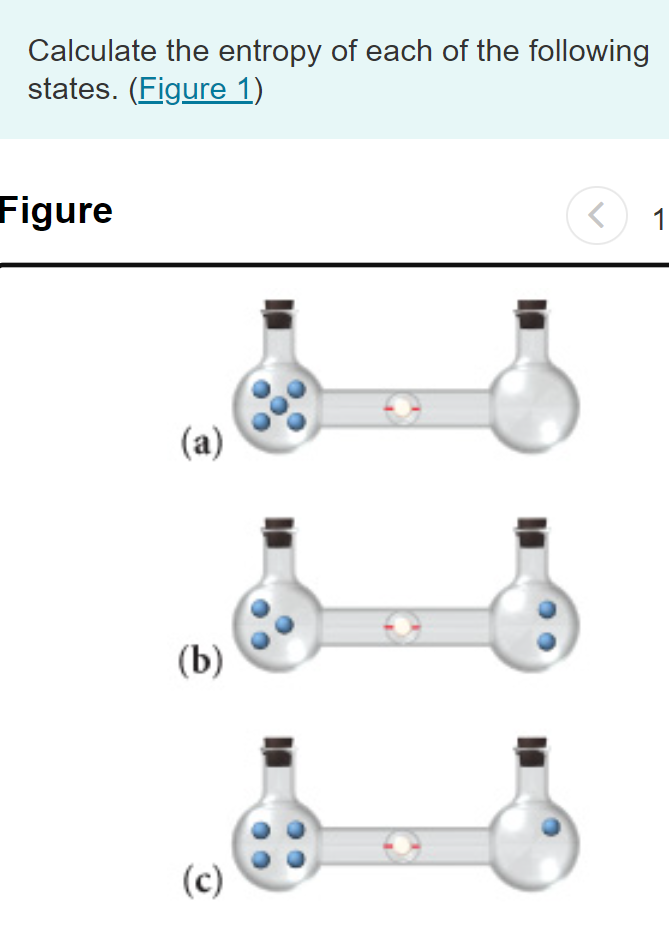
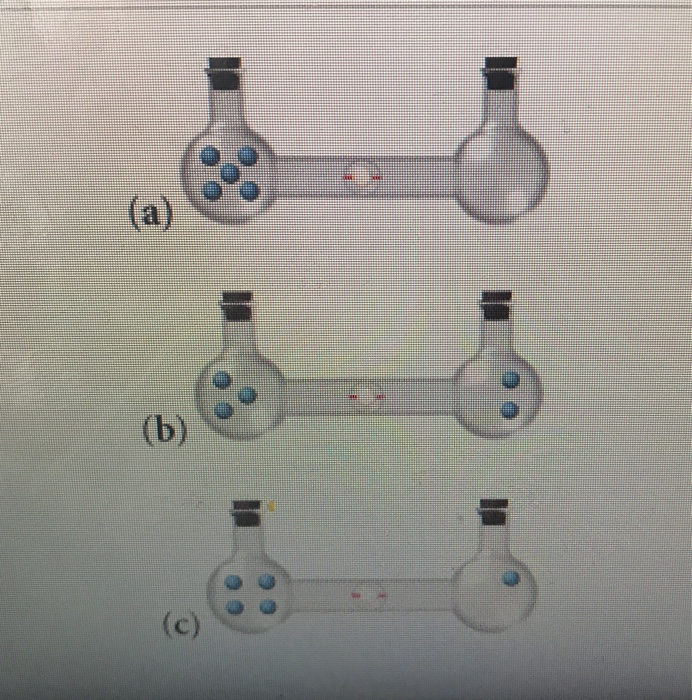
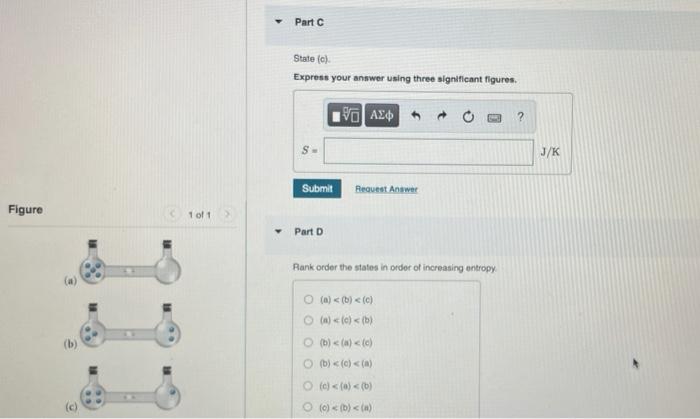
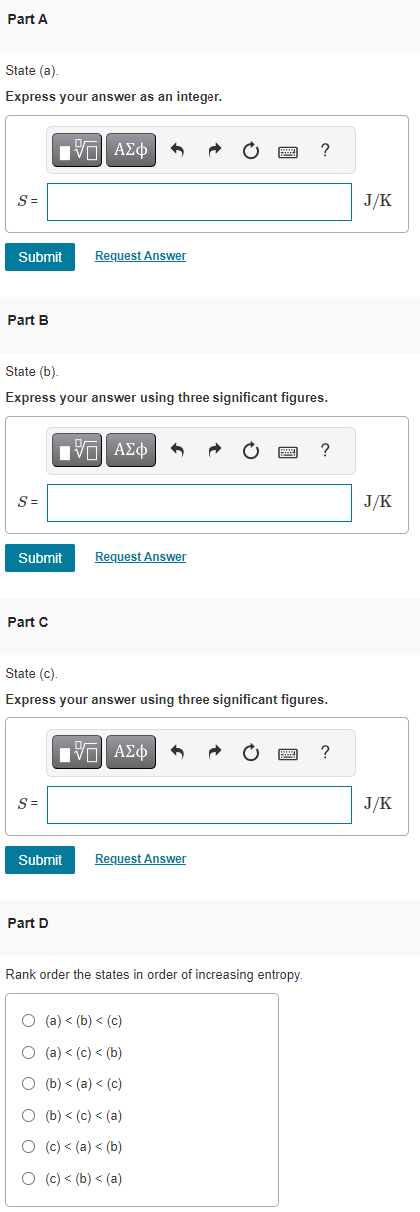
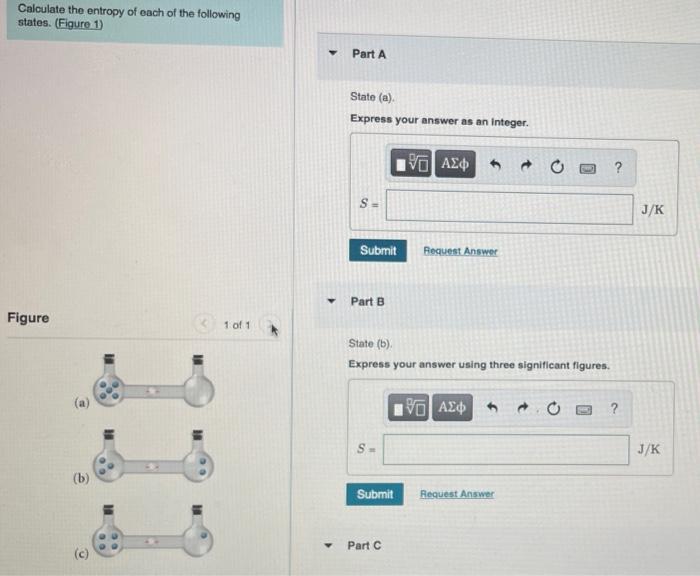
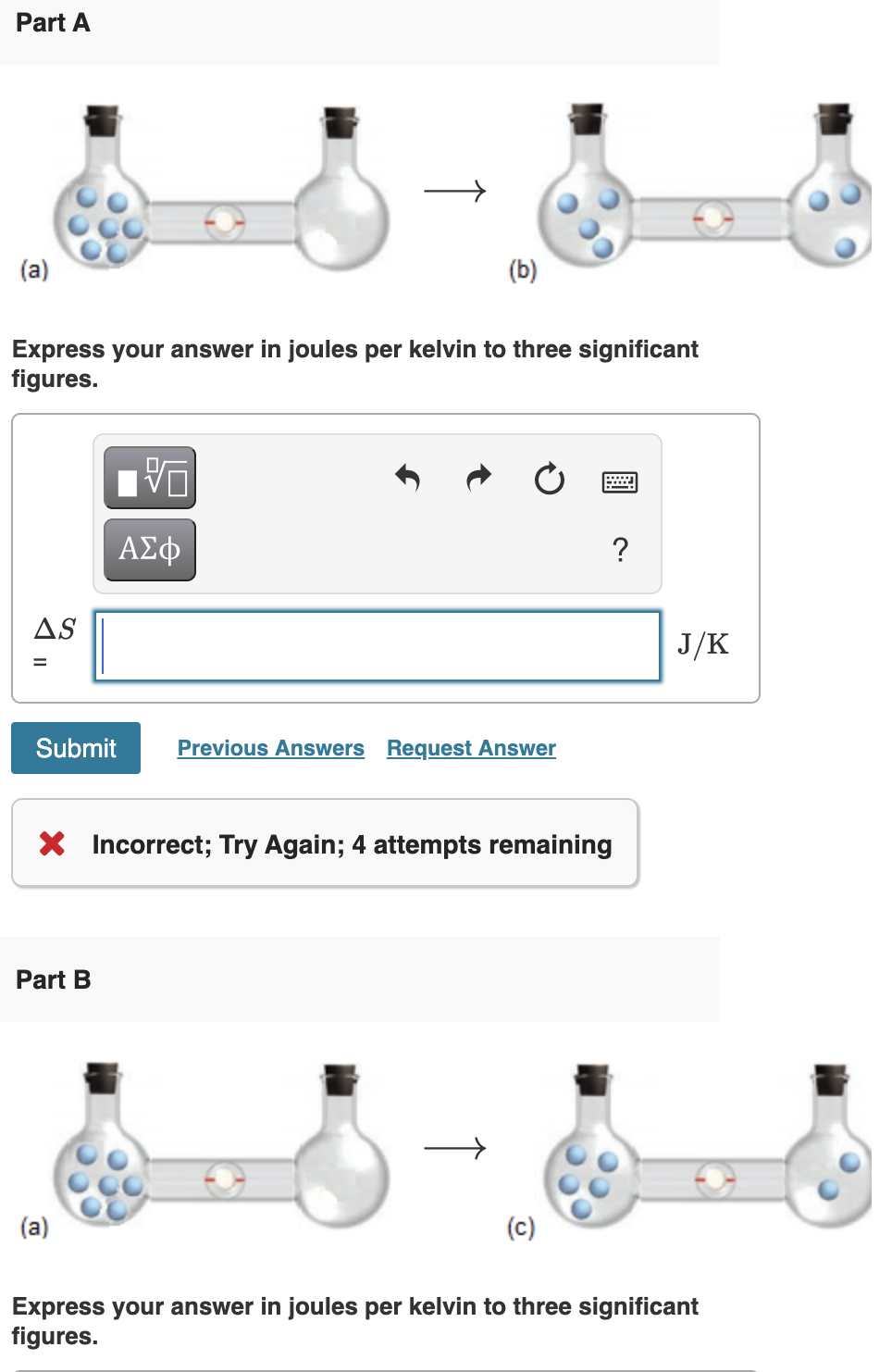
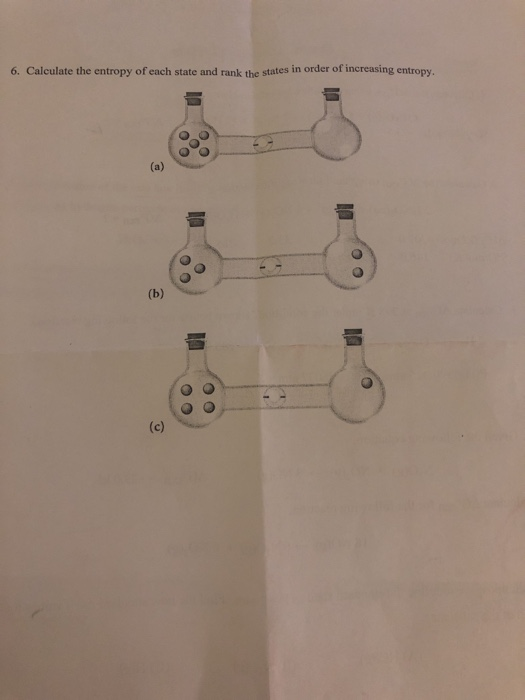
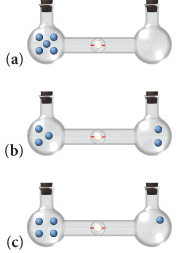
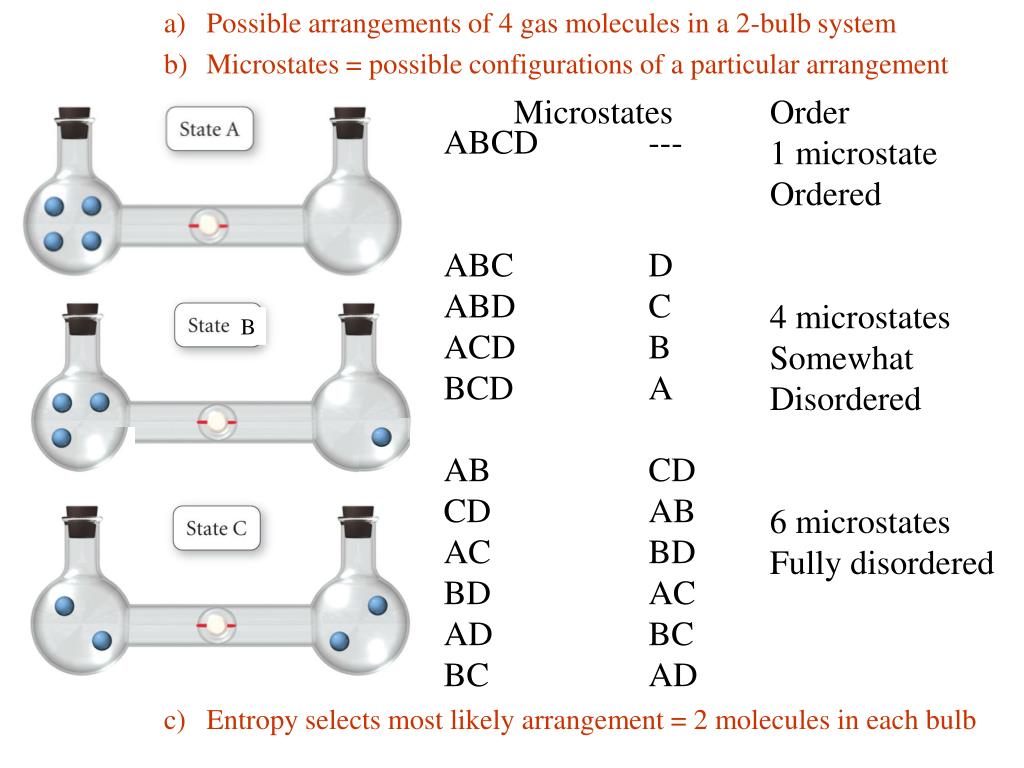
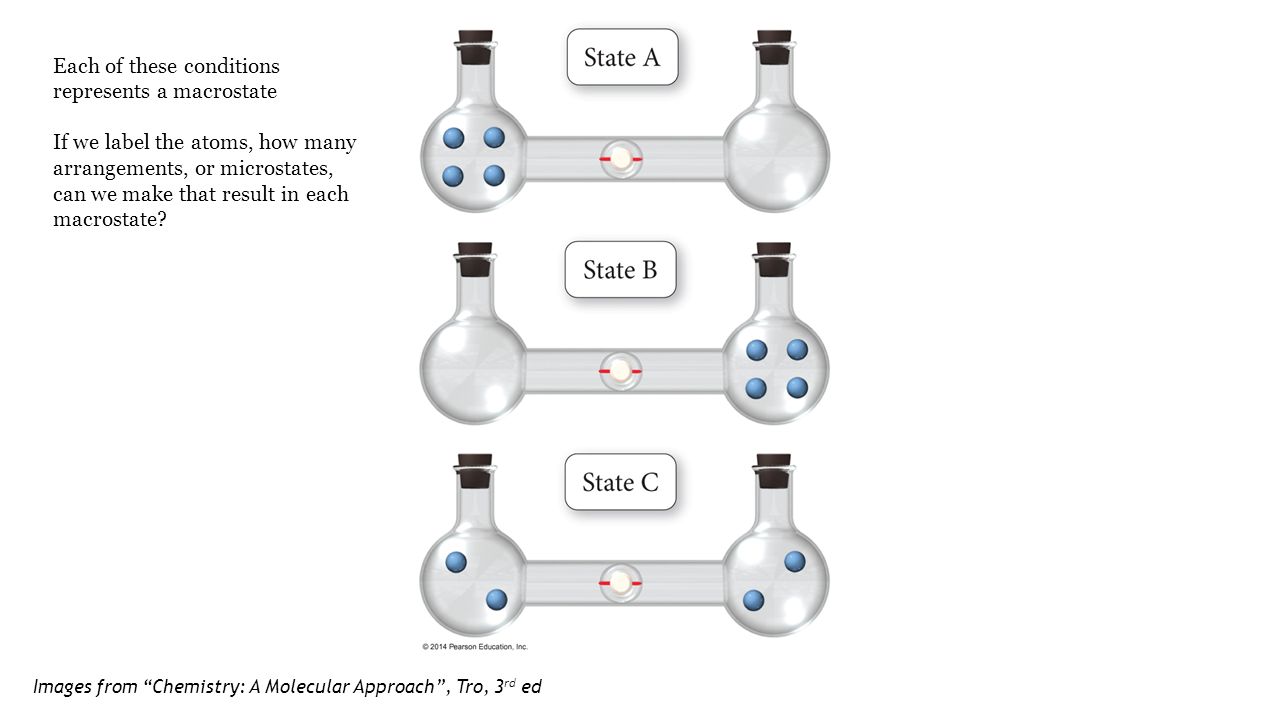
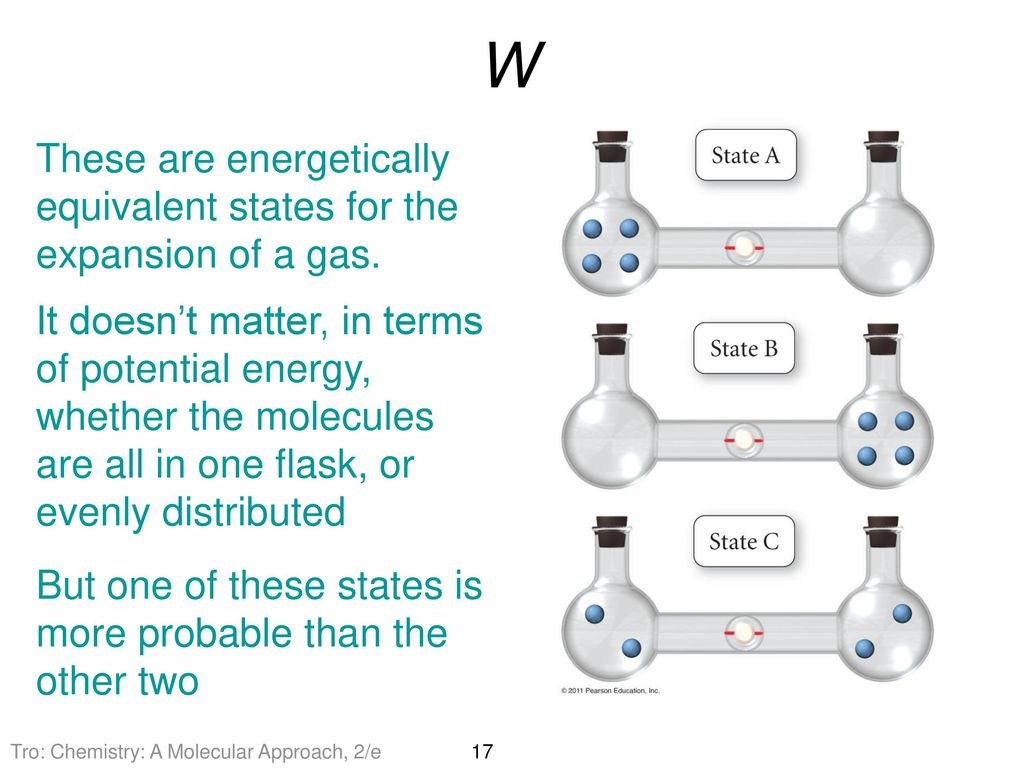
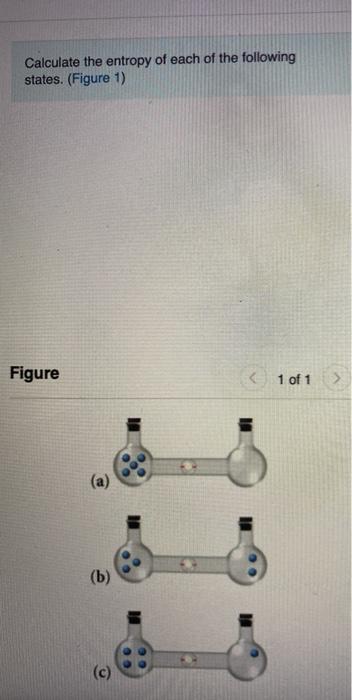
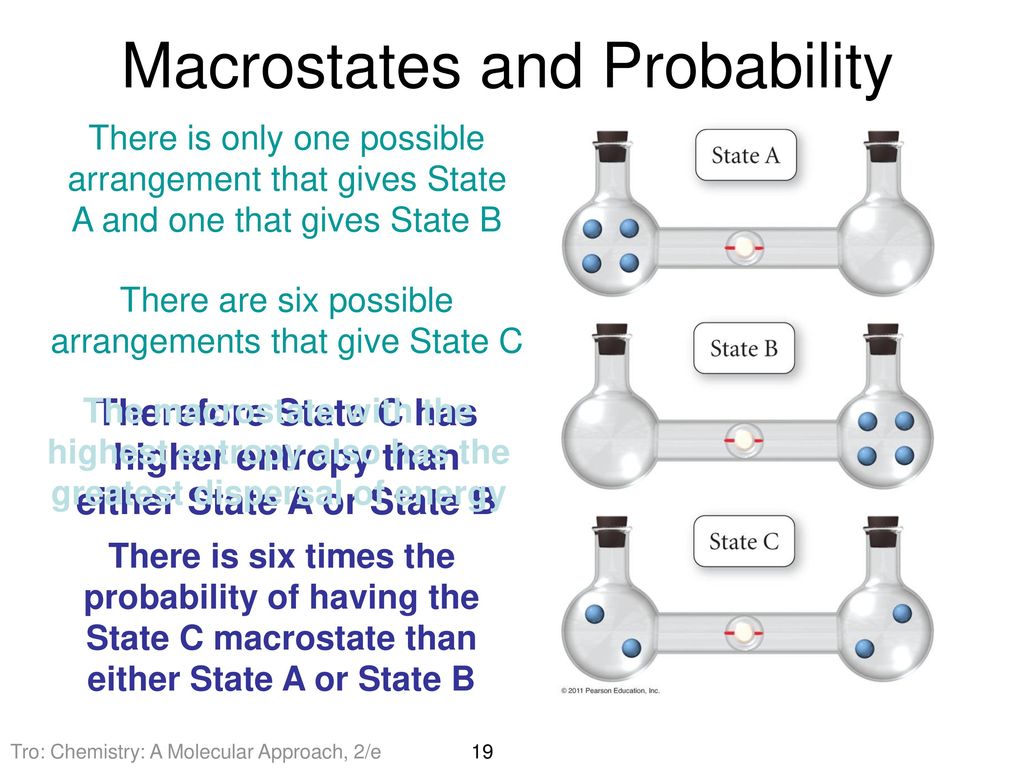
SOLVED:Calculate the entropy of each state and rank the states in order of increasing entropy. a. b. c.