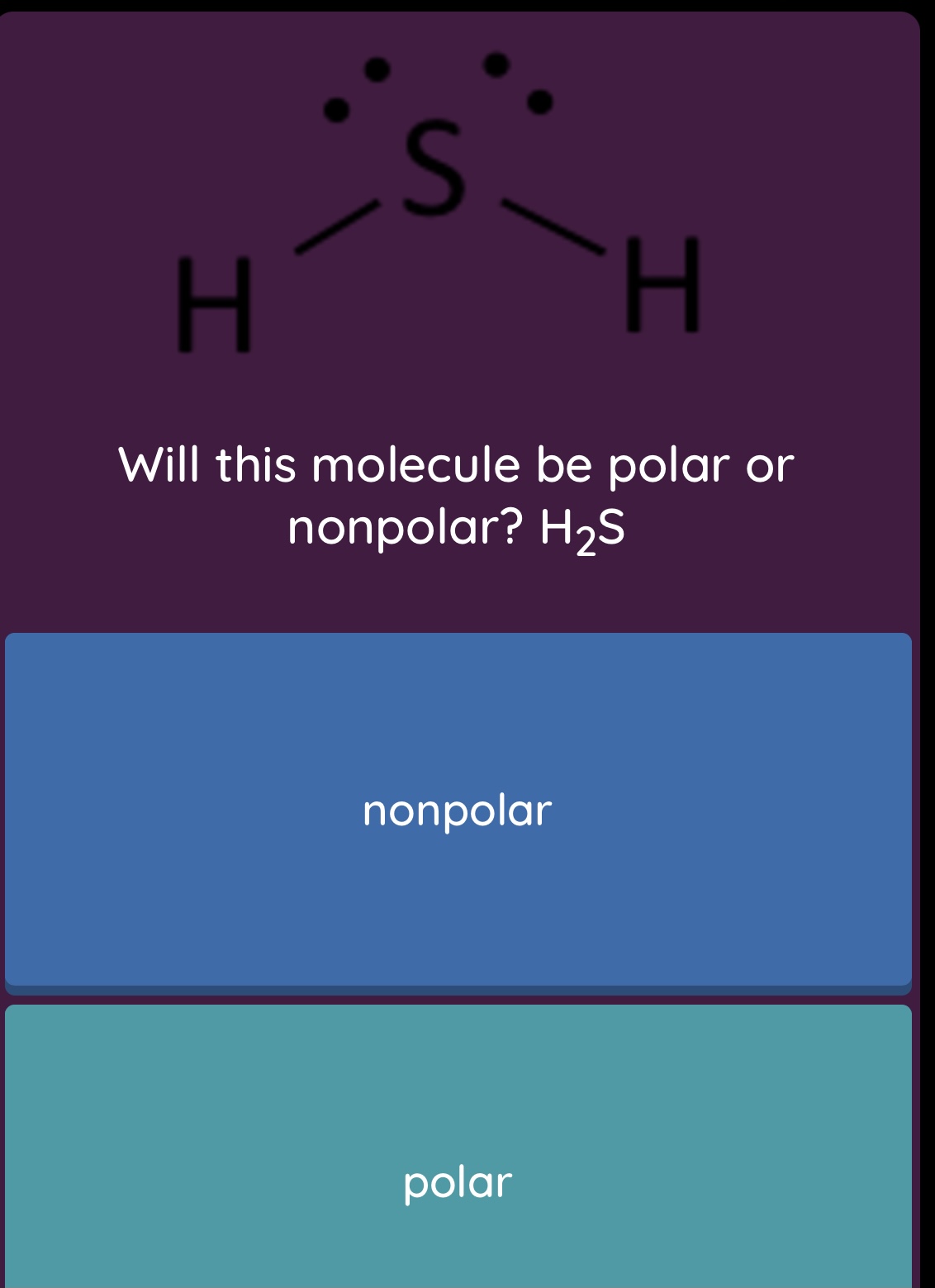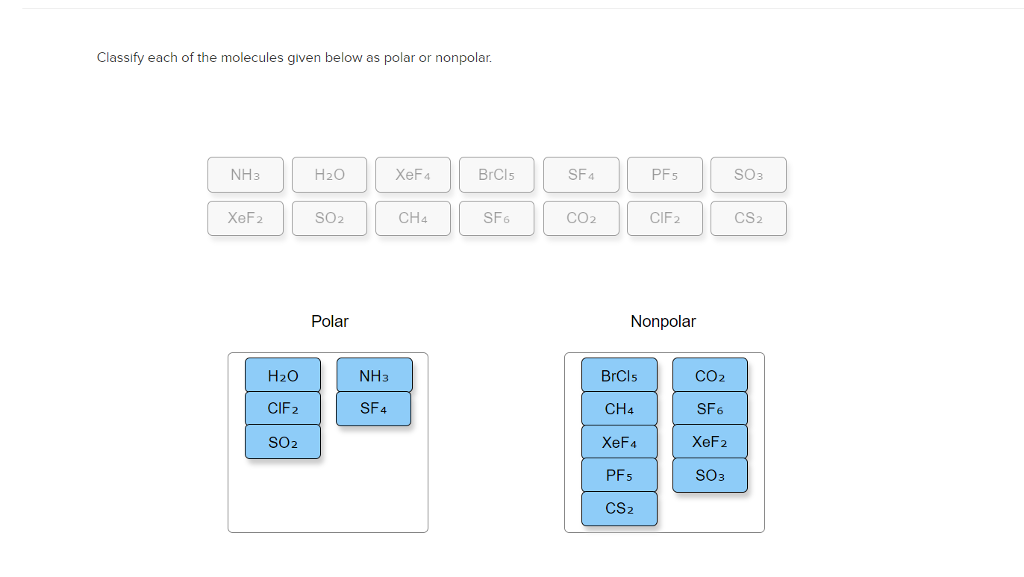
SOLVED: For each of the following molecules: (i) draw the correct Lewis structure; (ii) determine the molecular geometry and the type of hybridization on the central atom, and (iii) predict whether the

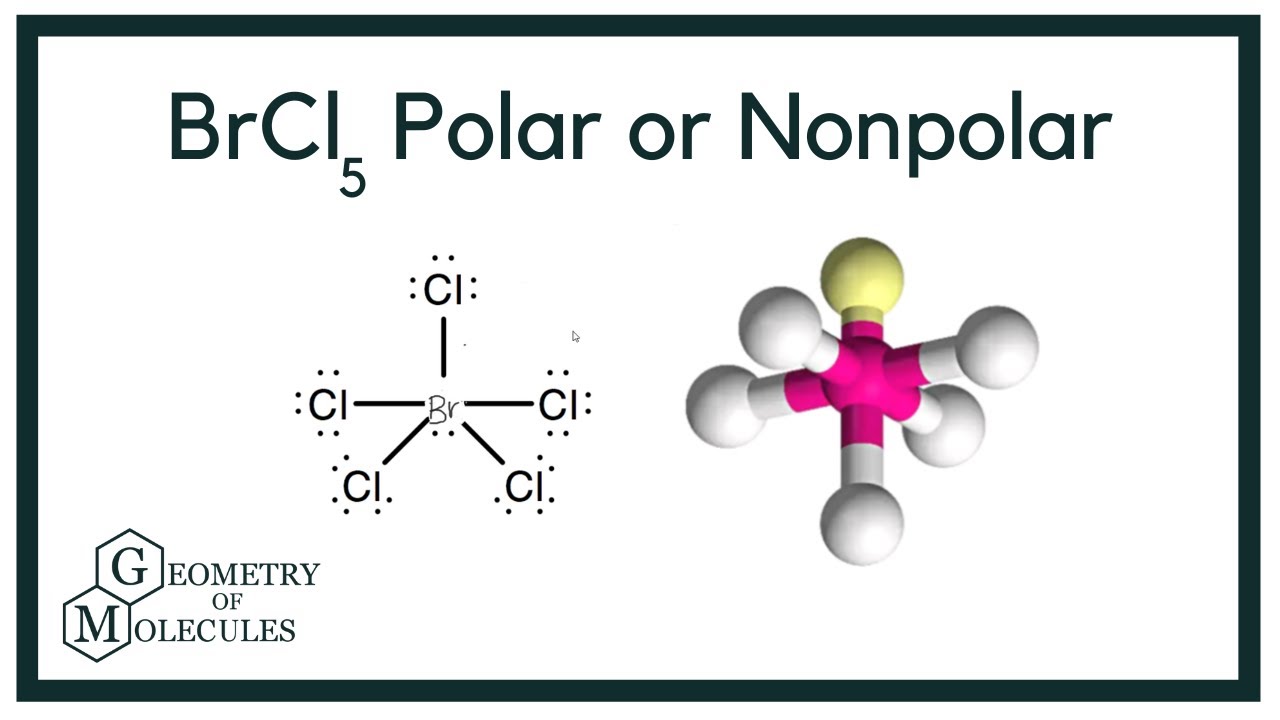
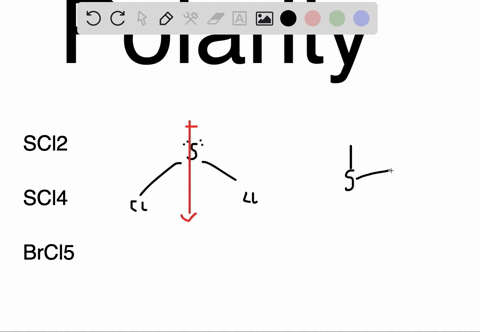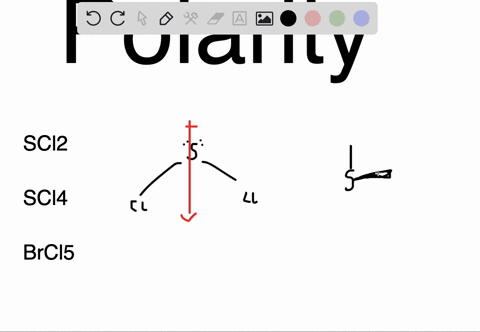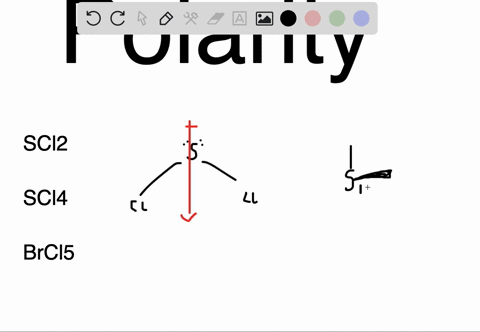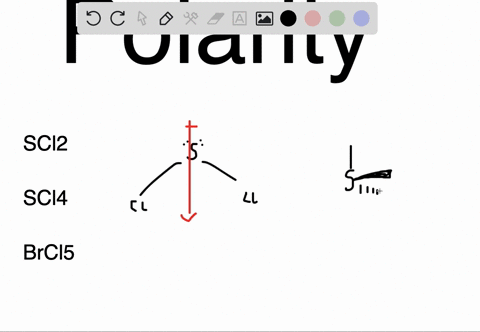
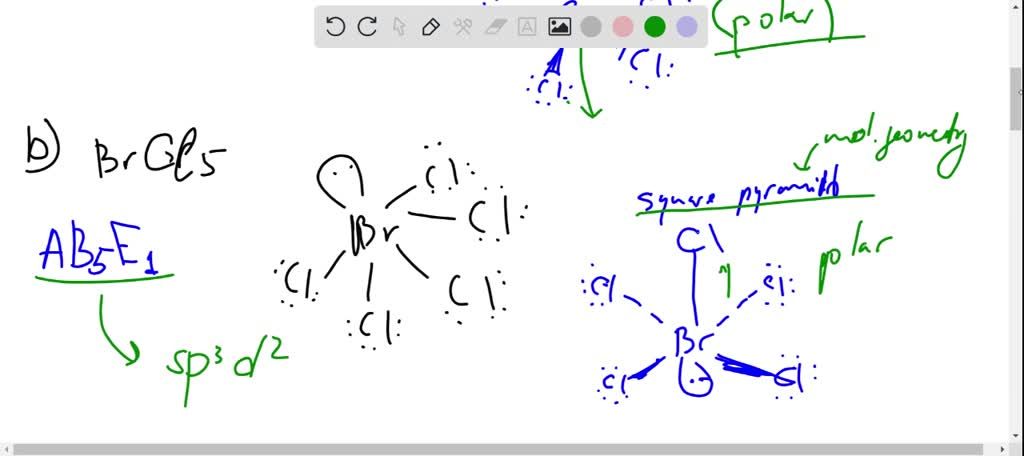
SOLVED: Determine whether each molecule or ion is polar or nonpolar. a. ClO3 - b. SCl2 c. SCl4 d. BrCl5
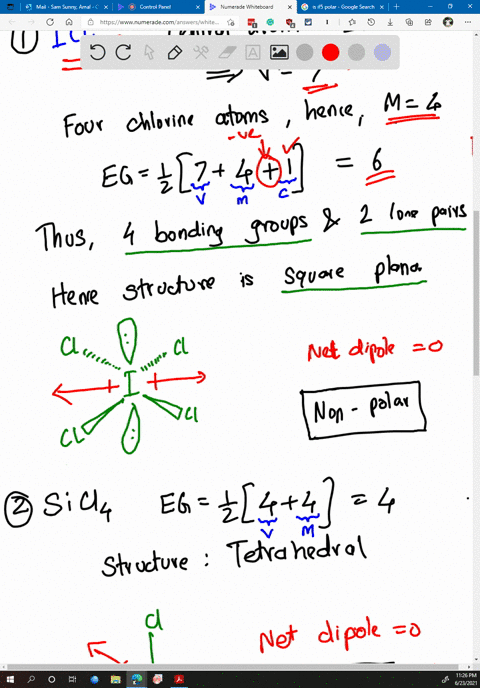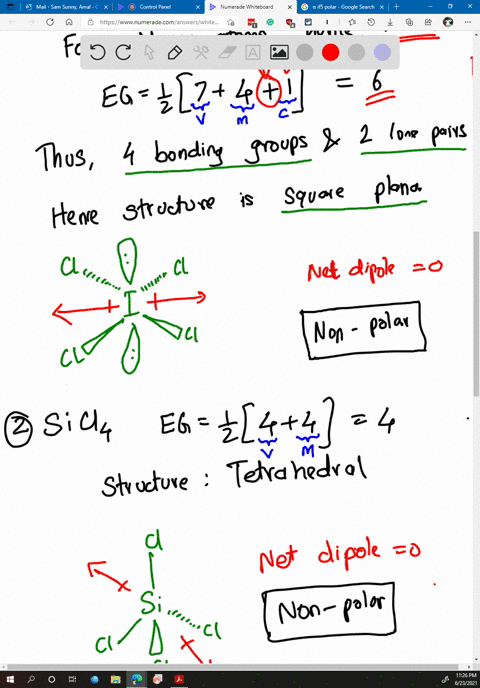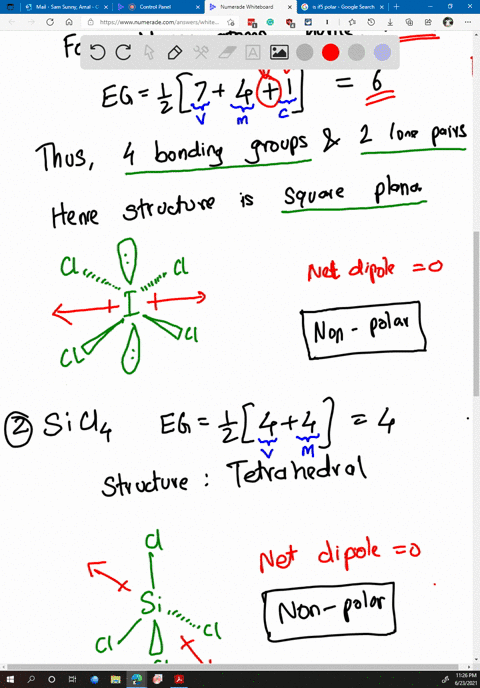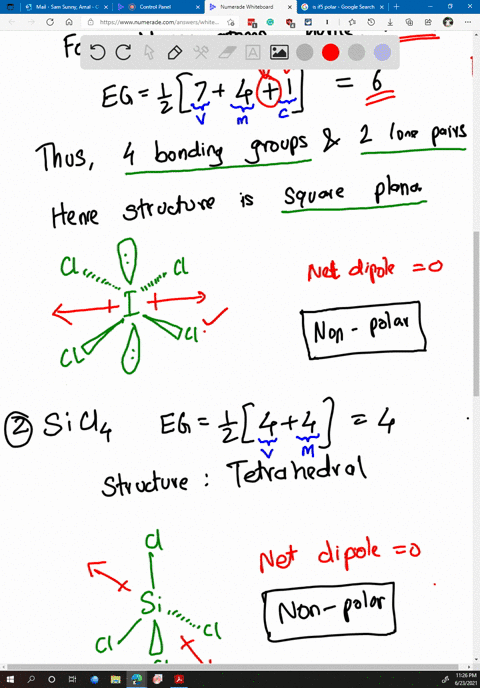
SOLVED: Determine whether each molecule or ion is polar or nonpolar. a. ClO3 - b. SCl2 c. SCl4 d. BrCl5